
60 Batches, Including Paracetamol and Cefixime, Fail CDSCO Quality Checks
In a recent drug safety update, 60 batches of medicines and medical devices have been classified as Not of Standard…

In a recent drug safety update, 60 batches of medicines and medical devices have been classified as Not of Standard…
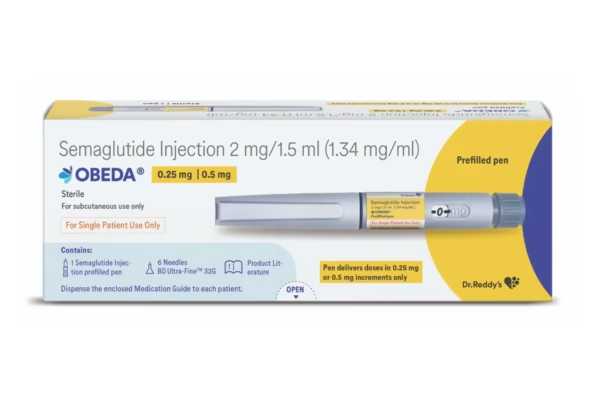
Dr. Reddy’s Laboratories has entered the fast-growing GLP-1 therapy segment with the launch of its semaglutide generic, Obeda, for Type…

Novartis AG’s move to exit its small Indian subsidiary once again highlights the limited appetite of global pharmaceutical giants to…

India is facing a rising incidence of metabolic, degenerative and autoimmune disorders, largely due to changing lifestyles and increasing environmental…

The detection of a counterfeit batch of Abhayrab, a rabies vaccine manufactured by government-owned Indian Immunologicals Limited (IIL), has highlighted…

AIIMS Delhi has conducted India’s first dedicated clinical trial of an advanced brain stent for the treatment of severe strokes,…

The Central Drugs Standard Control Organisation (CDSCO) has initiated a 100-day nationwide awareness campaign highlighting the recent reduction in Goods…

The World Health Organisation said it is closely monitoring the situation in India following the deaths of 20 children in…
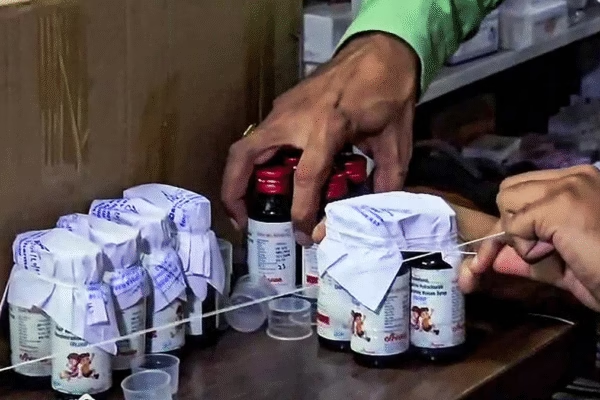
The Uttar Pradesh government on Sunday (Oct 5) banned the sale, import, and export of Coldrif cough syrup following reports…
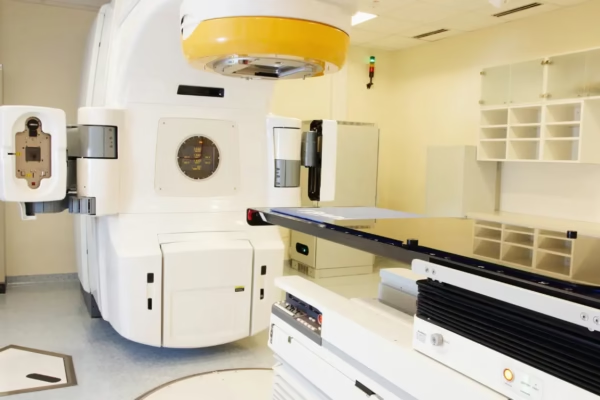
In a key development for the medical device industry, Andhra Pradesh now has its first Central Drugs Standard Control Organisation…