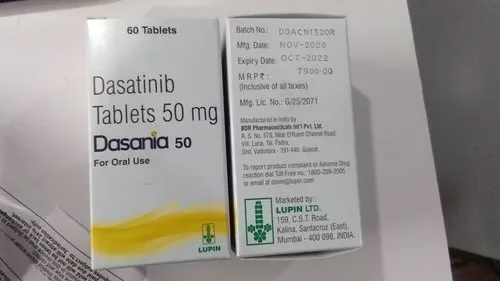
Lupin Limited has announced the launch of Dasatinib tablets in the United States after receiving approval from the US Food and Drug Administration for its Abbreviated New Drug Application (ANDA). The product has been developed in collaboration with Pharmascience Inc.
The Dasatinib tablets will be available in multiple strengths 20 mg, 50 mg, 70 mg, 80 mg, 100 mg and 140 mg and are therapeutically equivalent to Sprycel tablets marketed by Bristol-Myers Squibb.
Dasatinib is indicated for the treatment of newly diagnosed adults with Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) in the chronic phase. It is also approved for adults with chronic, accelerated or myeloid/lymphoid blast phase Ph+ CML who are resistant or intolerant to prior therapies, including imatinib. Additionally, the drug is indicated for adults with Ph+ acute lymphoblastic leukemia (ALL) who have failed or are intolerant to previous treatment.
The approval further extends to pediatric patients aged one year and above with Ph+ CML in the chronic phase, as well as children aged one year and older with newly diagnosed Ph+ ALL when used in combination with chemotherapy.
According to IQVIA data (MAT October 2025), the reference listed drug, Sprycel, recorded estimated annual sales of approximately USD 930 million in the US market.
Mumbai-based Lupin is a global pharmaceutical company with a presence in over 100 countries. It has a strong portfolio spanning branded and generic formulations, complex generics, biotechnology products and active pharmaceutical ingredients. Lupin operates 15 manufacturing facilities and seven research centers worldwide and employs more than 24,000 people, with major operations in India and the US across several therapeutic segments.
Pharmascience Inc., headquartered in Montreal, is among Canada’s largest pharmaceutical manufacturers. The company supplies high-quality medicines to over 50 countries and is known for its agile development capabilities and Canadian-based manufacturing, supporting healthcare systems with reliable and accessible therapies.