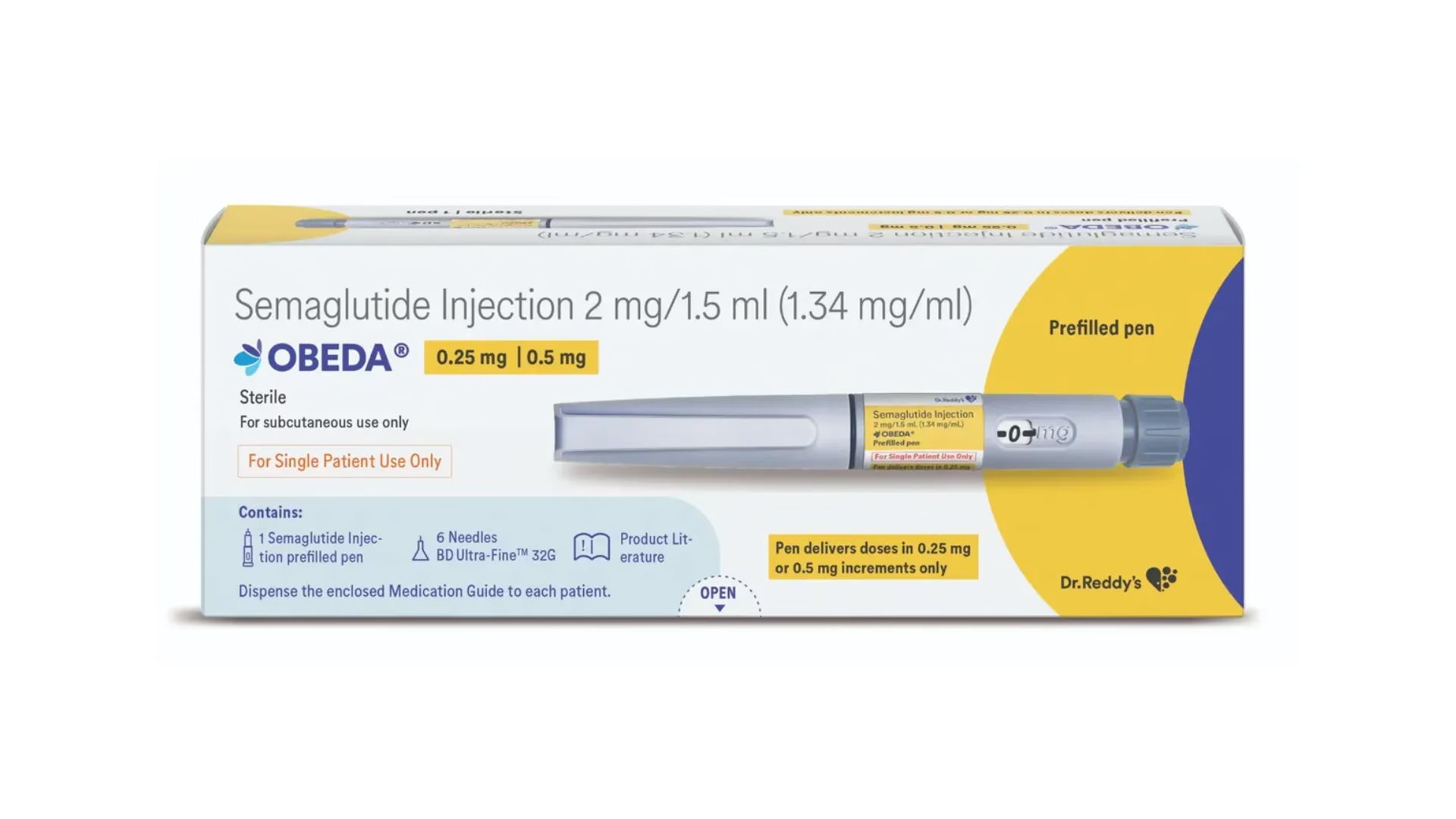
Dr. Reddy’s Laboratories has entered the fast-growing GLP-1 therapy segment with the launch of its semaglutide generic, Obeda, for Type 2 diabetes in India’s ₹1,446 crore market.
The launch comes immediately after the patent expiry of semaglutide, enabling wider generic competition. Priced at ₹4,200 per month for 2 mg and 4 mg doses, Obeda is about 62% cheaper than the highest-dose version of Ozempic from Novo Nordisk.
GLP-1 drugs help regulate blood sugar, promote satiety, and slow digestion, supporting both diabetes control and weight loss. Obeda will be available in a once-weekly, pre-filled injection pen, with each pen providing at least four doses.
The company plans to sell 12 million pens in its first year and aims to become one of the top five players in India’s semaglutide market. It is also awaiting approval from the Central Drugs Standard Control Organisation for a weight management version of the drug.
CEO Erez Israeli said this launch is part of a broader GLP-1 pipeline rollout, while executive M V Ramana highlighted global expansion plans. Canada remains a key market, though approval is currently on hold due to regulatory queries.
With in-house API and formulation capabilities, the company expects strong control over supply and quality. It is also exploring partnerships to expand branding and global reach.
India’s large diabetic population over 101 million, along with 136 million prediabetic individuals positions the country as a major growth market. Sales of injectable GLP-1 drugs have already surged 177% year-on-year, and with over 50 semaglutide generics expected soon, competition is set to intensify further.